Lauren Howell, PharmD
After rejecting approval twice, FDA has approved Furoscix (ScPharmaceuticals), a therapy to treat heart failure that is administered by an on-body infusor. With more than 7 million heart failure patients in the United States, there is now an option for them to self-administer this treatment from the convenience of their own home.
Recommended dosage and how it works
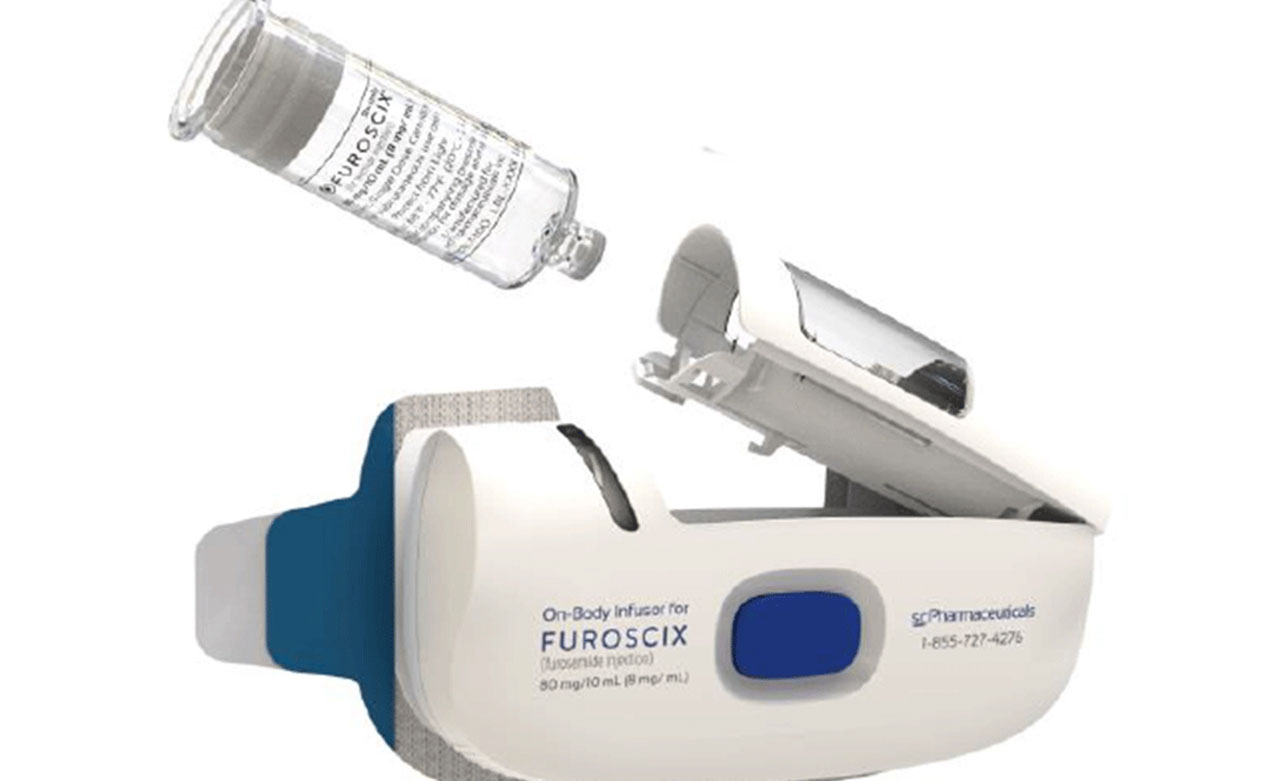
Furoscix uses an on-body infusor to subcutaneously administer furosemide injection. It is indicated for the treatment of congestion due to fluid overload in adults with NYHA Class II/III chronic heart failure. Furoscix is not indicated for emergency situations or in patients with acute pulmonary edema. Additionally, it is not intended to be used chronically and patients should be converted to an oral diuretic as soon as it is safe and practical to do so.
The single use, on-body infusor is preprogrammed to deliver 30 mg of Furoscix over the first hour and then 12.5 mg per hour for the subsequent 4 hours. The injection is packaged in a single-dose prefilled cartridge that contains 80 mg per 10 mL and is copackaged with a single use on-body infusor. Furosemide acts by inhibiting the reabsorption of sodium and chloride in the proximal and distal tubules as well as in the loop of Henle, causing a high degree of diuresis.
Drug interactions
Furoscix should be avoided in combination with aminoglycoside antibiotics as there is an increased potential for ototoxicity when these medications are used together. Additionally, Furoscix should be avoided in combination with ethacrynic acid due to the risk of ototoxicity. There is a risk of salicylate toxicity when used in combination with salicylates.
If Furoscix is taken in combination with cisplatin and nephrotoxic drugs, there is a risk of both ototoxicity and nephrotoxicity. Coadministration with lithium can increase the risk of lithium toxicity. There is increased risk of hypotension and renal failure if Furoscix is administered with renin-angiotensin inhibitors.
Combination with adrenergic blocking drugs carries the risk of potentiation. Lastly, if Furoscix is used alongside drugs undergoing renal tubular secretion, there is a risk of toxicity potentiation.
Adverse effects and contraindications
Furoscix is contraindicated in patients with anuria, hypersensitivity to furosemide or medical adhesives, and hepatic cirrhosis or ascites. The most common adverse reactions during treatment with the Furoscix infusor were administration site and skin reactions such as erythema, bruising, edema, and infusion site pain. Patients should be monitored for fluid, electrolyte, and metabolic abnormalities.
Additionally, patients should be monitored for dehydration. Avoid higher than recommended doses of Furoscix and monitor for ototoxicity. Acute urinary retention has occurred in some cases so patients should be monitored for symptoms of urinary retention.
Patient counseling
Patients should be counseled on the potential for postural hypotension that sometimes occurs and informed that this can usually be managed by getting up slowly. Patients should be advised to let their health care provider know of any supplements that they are taking. Potassium supplements may be necessary to avoid hypokalemia.
Patients with diabetes should be notified that furosemide may increase blood glucose levels. Patients should also be advised that there is the potential for photosensitivity while taking furosemide. Hypertensive patients should be counseled on avoiding medications that may increase blood pressure such as OTC products for appetite suppression and cold symptoms.
Provide patients with administration instructions and counsel them on how to use the on-body infusor. It is important to note that Furoscix is intended for use in a setting where the patient can limit their activity for the duration of administration. Patients should inspect the prefilled cartridge prior to administration and should not use the Furoscix if the solution is discolored or cloudy. Furoscix should be a clear to slightly yellow color.
The prefilled cartridge should be loaded into the on-body infusor, and the cartridge holder should be closed. Then the patient should peel away the adhesive liner on the on-body infusor and apply onto a clean, dry area of the abdomen between the top of the beltline and the bottom of the ribcage that is not tender, bruised, red, or indurated. The distance from the top of the beltline to the bottom of the ribcage should be at least 2 ½ inches.
The injection is started by firmly pressing and releasing the blue start button. The patient should not remove the infusor until the injection is complete, which is signaled by a solid green status light and beeping sound. ■